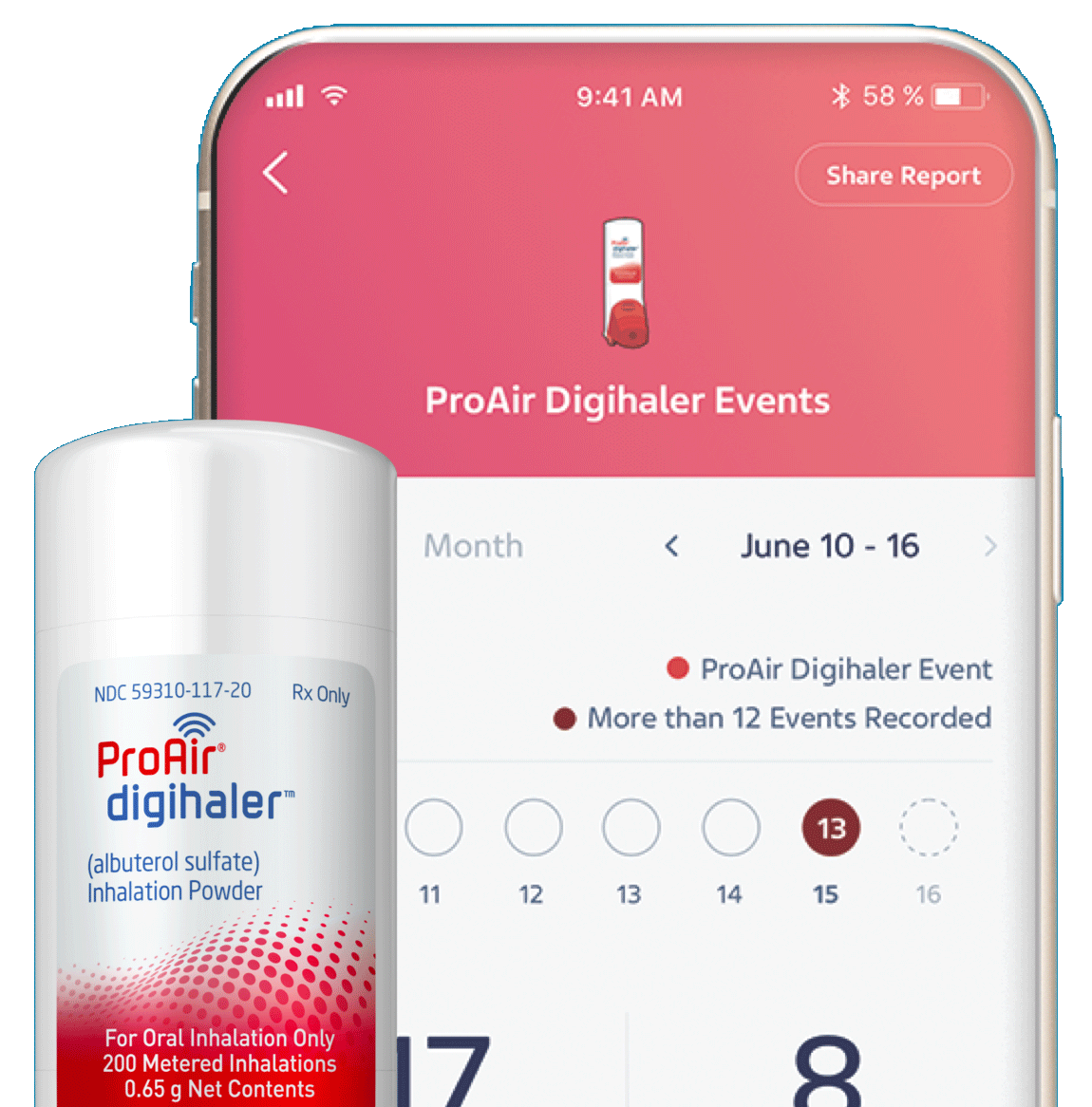
ProAir Digihaler (Albuterol Sulfate) Inhalation Aerosol Inhaler 90mcg — Mountainside Medical Equipment

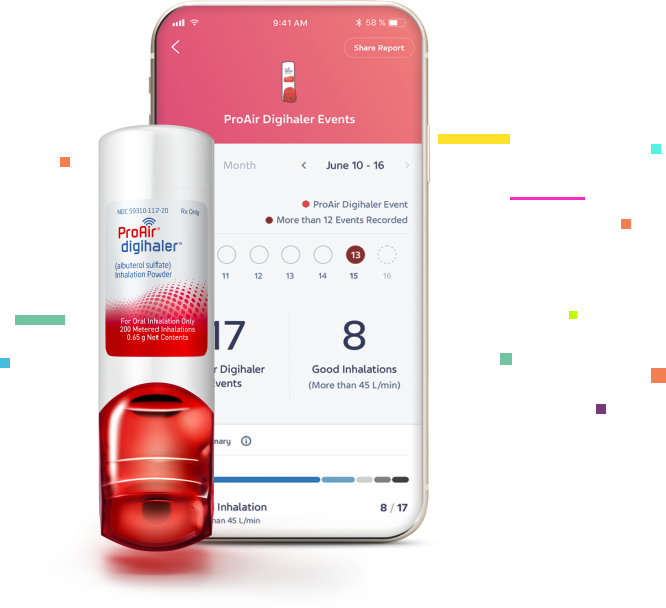
Teva to Present First Data Set from the CONNECT1 Clinical Trial on ProAir® Digihaler® (albuterol sulfate) Inhalation Powder at the European Respiratory Society (ERS) International Congress 2022

Teva Launches Two Digital Inhalers in the U.S., AirDuo® Digihaler® (fluticasone propionate and salmeterol) Inhalation Powder and ArmonAir® Digihaler® (fluticasone propionate) Inhalation Powder | Business Wire

ProAir RespiClick (albuterol sulfate) Inhalation Powder, 200 Metered Inhalations, Teva Pharmaceuticals USA (RX) Ingredients and Reviews

Teva Announces FDA Approval of First and Only Digital Inhaler with Built-In Sensors – ProAir® Digihaler™ | SnackSafely.com