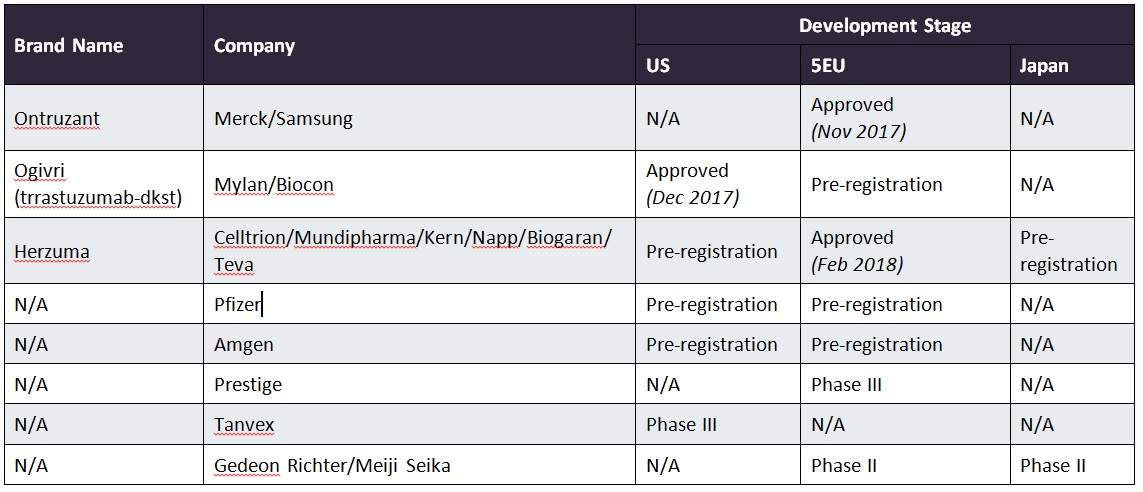
Teva and Celltrion Healthcare Announce the Launch of TRUXIMA® (rituximab-abbs) Injection for Rheumatoid Arthritis, the Only Biosimilar to Rituxan® (ri

Say hello to Roche's worst-case scenario: Teva's Rituxan biosim set to launch in U.S. | Fierce Pharma

JUNG, Ji-Ho on Twitter: "#Ajovy approved by FDA. #teva sells #Celltrion will make it in Korea. https://t.co/tKTp91jDXs" / Twitter