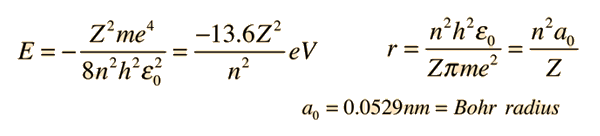1. The first Bohr's radius (for electron in hydrogen atom in the ground state): 2. The ground energy level in hydrogen atom: - ppt video online download

10. The value of Bohr's radius of hydrogen atom is : 0.529 x 10 cm 60.529 x 10-10 cm c) 0.529 x 10-12 cm d) 0.529 x 10-6 cm

Infinite Square Well “Particle in a Box”: Hydrogen (like) Atom Bohr Model: eV Bohr radius: a 0 = nm Orbital angular momentum. - ppt download

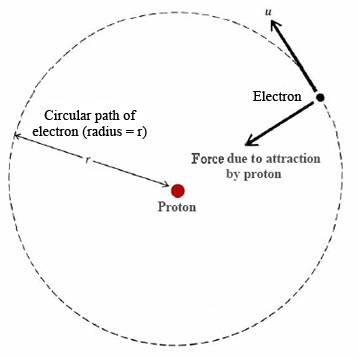
Using Bohr's postulates of atomic model, derive the expression for radius of nth electron orbit. Hence, obtain the expression for Bohr's radius. | Snapsolve

Which hydrogen like species will have same radius as that of Bohr orbit of hydrogen atom ? | Target Batch

Bohr radius of small hydrogen atom as a function of orbital quantum... | Download Scientific Diagram
What is the most possible radius (in PM) for an electron in the first orbit of a hydrogen atom? - Quora

1. The first Bohr's radius (for electron in hydrogen atom in the ground state): 2. The ground energy level in hydrogen atom: - ppt video online download
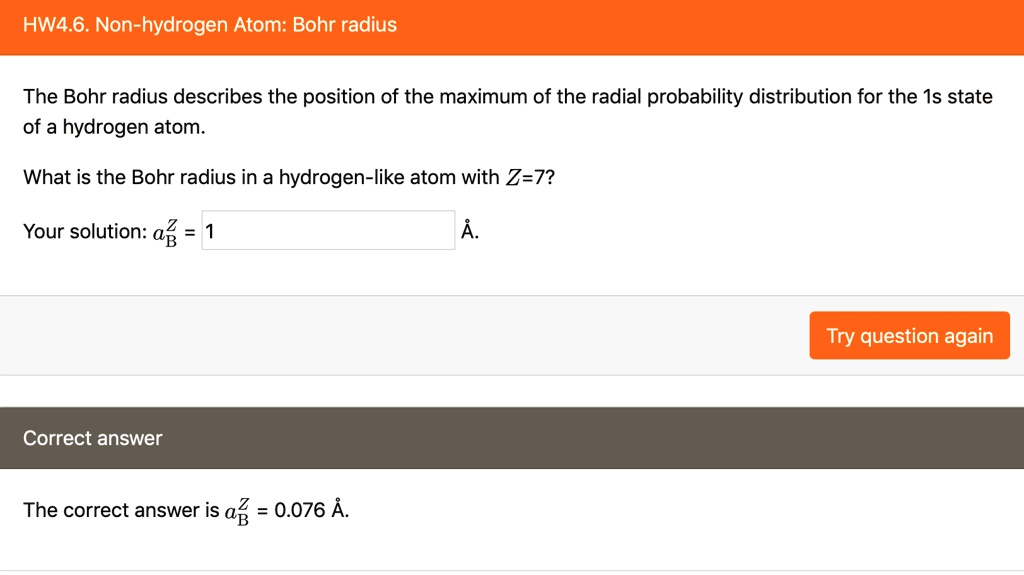
SOLVED:HW4.6. Non-hydrogen Atom: Bohr radius The Bohr radius describes the position of the maximum of the radial probability distribution for the Is state of a hydrogen atom_ What is the Bohr radius

OpenStax College Physics Solution, Chapter 30, Problem 13 (Problems & Exercises) | OpenStax College Physics Answers

MathType on Twitter: "The most probable distance between the nucleus and the electron in a hydrogen atom in its ground state is given by the Bohr Radius. This physical constant is named

Obtain the first Bohr radius and the ground state energy of a muonic hydrogen atom (i.e., an ato... - YouTube
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community